 Next Month: Monday, March 21st Dinner Meeting
Next Month: Monday, March 21st Dinner Meeting The FDA Drug Approval Process
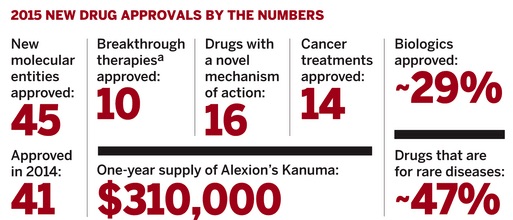
Source: Chemical & Engineering News, Feb. 1, 2016 issue
Speaker: • Clif Hotvedt, Vice President, Global Director, Medical & Scientific Affairs, Ketchum, Inc. Bio.
The U.S. Food and Drug Administration (FDA) traces its roots back to the Import Drugs Act of 1848, passed as a result of the United States at that time being, “the world's dumping ground for counterfeit, contaminated, diluted, and decomposed drug materials.” While it was at first only mandated that manufacturers demonstrate that drugs were safe, regulation over time required that proof of effectiveness was also important.
Many new regulations were passed in response to public health disasters. More recent changes to the FDA review process allow for getting drugs to the market sooner. The FDA’s mantra is that no drug is entirely safe and no drug is entirely effective. The balance between these two needs is constantly being challenged.
This presentation will review how the FDA’s approach to drug regulation has evolved from crises such as thalidomide babies through current issues such as personalized medicine, generic drugs and biosimilars and emerging infectious diseases.
When: Monday, March 21st, 5:30 - 7:30 pm (5:30-6:15 pm: Registration, Networking & Buffet Dinner; 6:15-7:30 pm: Program)
Where: Pfizer Building, 219 East 42nd Street, Manhattan. Map
Costs: for Networking, Buffet, and Program. • Members of AIChE, Metro NY Section: $25; • Non- Members: $35; • Students (Undergraduates): Free for first 20 undergrads, who register by Friday 3/11; $5 afterwards. You must register; • Graduate Students: $10.
Questions: Contact David Deutsch at info@aiche-metrony.org or call (917) 684-1659.
* * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * *
If you are not a Member of this Section, you can apply the $10 additional amount paid as a Guest to a full Membership for 2016. Please notify Treasurer Joel Kirman that you wish to be a Member. Register Now.
_______________________________________________________________
Save the Dates: 2016 Meeting Dates
2016 Dates Details to be determined for Spring Dinner Meetings.
• Tuesday, February 16th - "Process Safety-It’s not just for Refiners and Chemical Companies" (Speaker: Richard W. Sarnie, CSP, P.E., ARM-E, CRIS, MLIS, Director, CCPS Projects, Center for Chemical Process Safety - AIChE)
• Monday, March 21st - "The FDA Approval Process" (Speaker: Clif Hotvedt, VP Global Director, Medical & Scientific Affairs, Ketchum, Inc.)
• Monday, April 18th
• Monday, May 16th
• Monday, June 20th
Where: Pfizer Building, 219 East 42nd Street, Manhattan. Map
_______________________________________________________________


